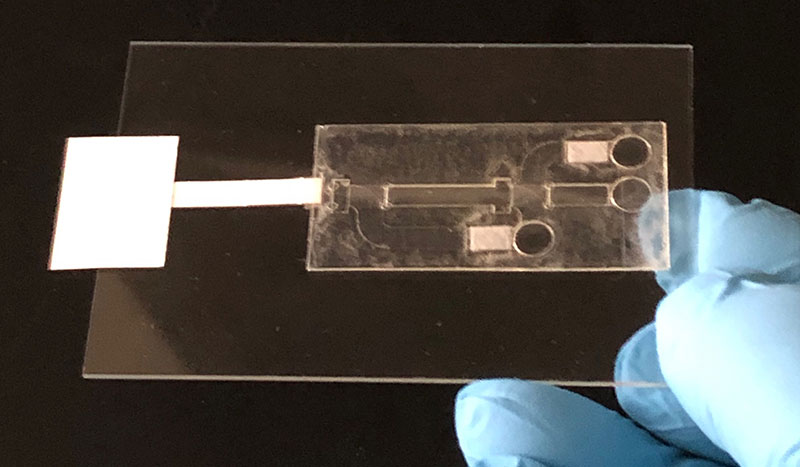
A prototype of the viral sensor CSU researchers have licensed to Quara Devices.
A small, inexpensive virus-detection technology invented by Colorado State University researchers will soon form the basis of a new product that could compete with standard diagnostic testing for COVID-19.
The new diagnostic device, which aims to be fast, portable and more accurate than currently available COVID-19 tests, is one of several technologies spawned from a collaboration among CSU researchers Brian Geiss, Chuck Henry and David Dandy. Combining their wide-ranging expertise in virology, chemistry and chemical engineering, the team has licensed their viral RNA-testing platform to Quara Devices, a startup company specializing in diagnostic biosensors.
The licensing deal, mediated by CSU Ventures, was inked earlier this summer and allows the company to move the invention into product engineering and design phases, while the CSU scientists continue testing the devices for efficacy and accuracy. The Chief Science Officer of Quara is Ken Reardon, a professor in the CSU Department of Chemical and Biological Engineering.
Geiss, Henry and Dandy have worked several years together developing low-cost biological diagnostic platforms for applications including viruses, bacteria and antibodies. For the device they’ve licensed to Quara, their original goal was detecting organisms with antimicrobial resistance. As COVID-19 was becoming a pandemic, they found they could generalize their basic platform, which they began developing over a year ago, into a sensitive test for RNA viruses, including coronaviruses like SARS-CoV-2.
Reardon, who joined Quara last year after consulting on a bacterial-detection technology for the company, approached his CSU colleagues about possibly extending their viral detection strategy into a product that Quara could commercialize. During these negotiations, the COVID-19 pandemic worsened, which further spurred the group toward a deal that would allow them to enter the COVID-19 diagnostics market. Their technology is envisioned as a point-of-need genetic analyzer for viruses and bacteria, including but not limited to SARS-CoV-2, the virus that causes COVID-19.
“This technology predated the pandemic, but because of its versatility as a platform, and the significant need for high accuracy in detecting COVD-19 in patients, we see an urgency to get it out now,” Reardon said. “We are trying to get this into the market within the year, which is a pretty aggressive acceleration.”
How it works
Henry, a professor in the Department of Chemistry, explained that the licensed technology is a paper-based, “lateral-flow” device reminiscent of a home pregnancy test. It recognizes a target sequence in the virus’s genetic material, then amplifies that signal to display a positive readout – like the line on a pregnancy test.
The test serves the same purpose as the genetic analysis technique called quantitative polymerase chain reaction (PCR), which is the gold standard for diagnosing COVID-19 from saliva samples. PCR requires large, expensive laboratory instrumentation and a “chain reaction” of copying the viral genome sequence of interest using temperature cycles. The CSU test works at one temperature, and it includes an extra chemical step that improves the test’s selectivity and accuracy. Because it combines all these elements in one handheld device, it doesn’t require laboratory analysis, keeping the cost down.
“I am very much a believer that if you don’t get a technology like this into someone’s hands who can make and produce it, it’s just an academic exercise,” said Henry, who has been involved in several CSU research-based startup companies. “It was a big deal for us to move this into a product that will help people.”
Beyond the genetic analyzer they’ve licensed, the team is continuing to develop other diagnostic tests with the same goals of accuracy, portability and cost effectiveness. For example, they are hard at work on a serological test that detects proteins, whether antibodies or antigens, that would compete with the industry standard “ELISA” tests used today.
Across disciplines
The research team first came together in 2015 under an Office of the Vice President for Research initiative called Catalyst for Innovative Partnerships that provided seed funding for cross-disciplinary work. CSU’s vice president for research office has for several years made strategic investments in research relating to outbreak diagnostics, including for the team that invented the Quara device, as well as another team that develops molecular tags for in vivo imaging and editing.

CSU researchers Chuck Henry, Brian Geiss and David Dandy.
Geiss is an associate professor in the Department of Microbiology, Immunology and Pathology with biological expertise in how RNA viruses replicate. Henry, an analytical chemist, has broad experience in microfabrication of paper-based chemical tests, chromatography and microfluidics. Dandy, a professor in the Department of Chemical and Biological Engineering, has expertise in transport mechanics, including the physics of how materials and fluids interact with sensor surfaces.
“All three of us work well together to cover all aspects of making biosensors and assay devices,” Henry said.
Rod Reum, CEO of Quara Devices, said: “We are excited to work with the high caliber cross-disciplinary CSU team, and for the great potential for this technology to be an accessible tool to quickly and accurately detect COVID-19 and other viruses.”