The devastation of mitochondrial diseases is felt by millions of people around the world, and about 1 in every 4,300 people in the United States.
The mechanics of these diseases, which can lead to poor growth, muscle weakness, neurological problems and more, lie in the genes inside mitochondria, which are the cellular organelles that produce the cell’s energy. Mitochondrial genomes, which are inherited only from the mother, are separate from the nuclear genomes we tend to think of and that are inherited equally from the mother and father.
In humans and other animals, mitochondrial genomes have extremely high mutation rates, and these mutations are passed from mother to child very easily. Scientists are eager to learn more about why these high mutation rates occur and what could be done to stop them, so that mitochondrial disease can become a thing of the past.
Colorado State University biologists are funded by the National Institutes of Health to seek answers to these very questions, but they’re not medical researchers – they’re plant biologists. It turns out humans have a lot to learn from plants, from a genetic standpoint, of how to keep our mitochondrial genomes free from mutations that cause disease.
How do plants experience mitochondrial mutations?
Amanda Broz, a research scientist in the lab of Dan Sloan, associate professor in the Department of Biology, led a recent study published in Proceedings of the National Academy of Sciences that sheds new light on how plants, however rarely, experience mutations in their mitochondrial genomes. Unlike humans, plants are able to quickly fix these mutations, and more importantly, not pass them on to their progeny.
In previous work, Sloan and his lab members hypothesized that genes responsible for DNA replication, recombination and repair in plant organelles like mitochondria and chloroplasts might be involved in keeping these organelles healthy and mutation-free. They honed in on a gene called MutS homolog 1, or MSH1 for short, which is found in plants but not humans. Generating plants in the lab with mutations in this gene, they found evidence that this gene is critical for keeping mitochondrial mutation rates low in plants.
A more detailed analysis followed, as they sought to understand how mutations in the genomes of both mitochondria and chloroplasts spread, both within the plant and across generations. What they found, and is detailed in their new paper, was that plants are very good at sorting normal (good) and mutant (diseased) DNA. Once the sorting process has happened, natural selection takes over: offspring that inherit diseased DNA are less likely to survive, so the mutation is not passed on to progeny.
In contrast, the diseased DNA in humans tends to stay mixed up with the good, and gets passed down through generations, because this sorting ability is much less effective. In more technical terms, plants remove heteroplasmy – the presence of both mutant and non-mutant DNA – from their cellular compartments much faster and more efficiently than their animal and human counterparts.
The researchers tracked the mitochondrial mutations they identified in a species called Arabidopsis over time and space using a sensitive technique called digital droplet PCR. This technique allowed them to analyze the amount of mutant versus normal mitochondrial DNA in these plants.
And, back to that special gene: Using the mutant plants they grew in the lab, the CSU biologists found that functional copies of MSH1 are what accelerate that DNA sorting process in plant mitochondria. They think that MSH1 is responsible for first identifying mutations in mitochondrial DNA, and fixing them. However, if a mutation persists for one reason or another, MSH1 can also work to quickly sort that mutation away from normal DNA.
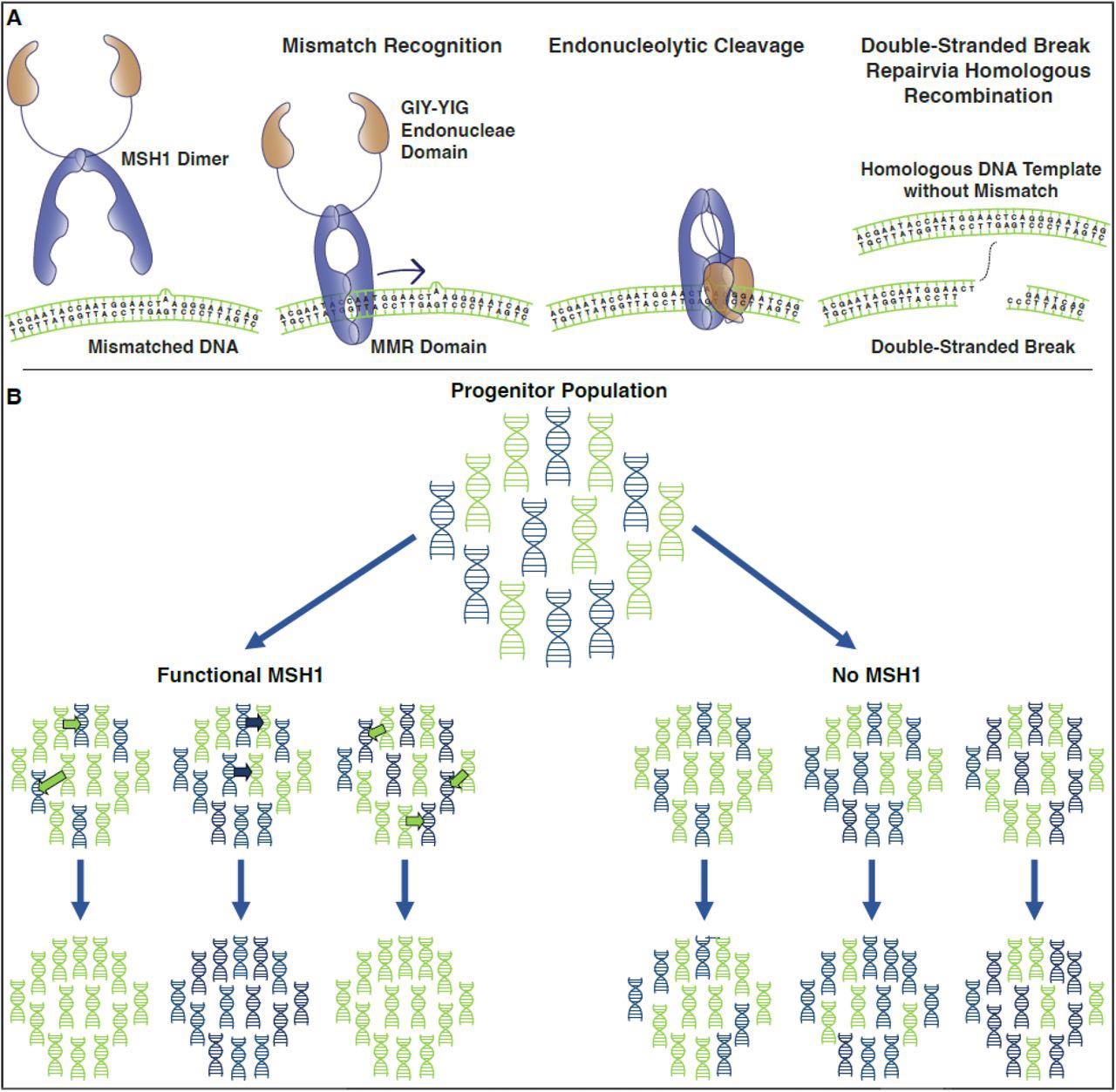
MSH1 repairs organelle DNA. The MSH1 protein slides along a DNA strand until it detects mutations. Then MSH1 cuts both strands of DNA. The DNA repair machinery in the cell uses homologous DNA (DNA of the same sequence) as a template to fix the mutation. B) MSH1 increases sorting of organelle DNA. A population of green (normal) and blue (mutant) organelle DNA molecules is shown. When functional MSH1 is present, it homogenizes copies of DNA, and the resulting populations contain either normal or mutant DNA. When MSH1 is absent, the DNA types remain mixed.
“Something that is really cool about our work is that it illustrates how nature has devised multiple ways of dealing with mutations in organelle genomes,” Broz said. “We know that mitochondrial mutations are one of the key factors leading to aging and disease in humans. Knowing how plants and other organisms maintain such low mitochondrial mutation rates can give us a better understanding of how this process goes awry in humans, and potentially how it can be remedied.”
The researchers have much more to learn from their mutant plants. They are next creating a mathematical model that attempts to understand what cellular forces are responsible for the different rates of organelle DNA sorting they observed in plants, and what role MSH1 plays in the process. That part of the project is being led by collaborators at the University of Bergen in Norway. They are also looking to investigate whether MSH1 has the same impact on sorting mitochondrial DNA in other plant types besides Arabidopsis, such as trees that have very different development patterns.
The Department of Biology is in the College of Natural Sciences at CSU.